- Valence Shell Electron Pair Repulsion – allows us to predict the shape of molecules to better learn their properties (they are 3-D versus 2-D Lewis structures)
- Molecules will assume arrangements that minimize the repulsion between the electron pairs around the central atom.
VSEPR Models: Two bonded pair

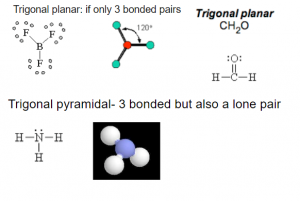
VSEPR Models: Three bonded pairs of electron
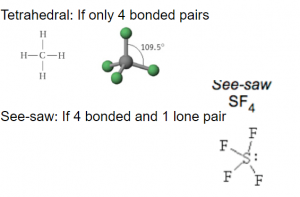
VSEPR: Four bonded pairs of electron
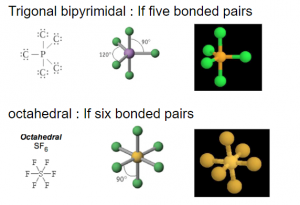
VSEPR: Five and Six bonded pairs of electron
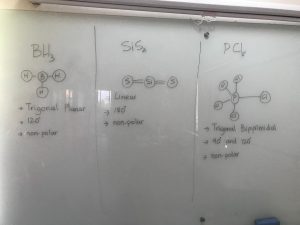
Polarity – based on the shape of the molecule
-
- In a polar molecule, there is a net unequal sharing of the electrons.
- If two dipoles are at angles they will NOT cancel out.
- In a non-polar molecule, there is a net equal sharing of electrons.
- If two dipoles are in opposite directions they will cancel each other.
- This can be determined by looking at electronegativity and shape.